Biotech support

Biotech Support
In order to promote the commercialization and industrialization of the regenerative medicine and cell medicine field, which is expected to grow significantly in the future, in addition to hands-on support by the incubation manager based on KSP Biotech Lab, "Regeneration and cell medicine industrialization promotion business" from Kanagawa Prefecture We are entrusted with the support through the "Regenerative medicine & Cell therapy industrialization network of Kanagawa (RINK)" and the "Regenerative Medicine and Cell Therapy Industrialization Joint Project".
Cooperation with Kanagawa Prefecture (operations taken on commission)
“Regenerative Medicine and Cell Therapy Industrialization Joint Project”
By supporting regenerative medicine and cell therapy projects (research, development and commercialization) conducted by start-ups etc. within the prefecture, Kanagawa Prefecture seeks to facilitate early and reliable commercialization within its borders and to create networking effects in related industries. Accordingly, with the aim of promoting regenerative medicine and cell therapy industrialization within the prefecture, it has rolled out operations for the promotion of industrialization of regenerative medicine and cell therapy.
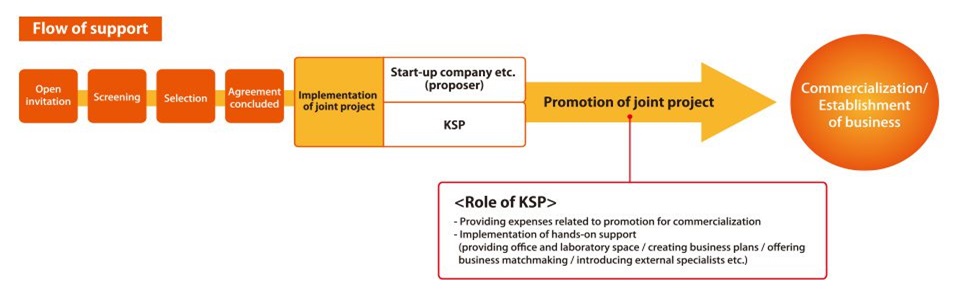
KSP has taken on these operations on commission, as part of which it has launched the Regenerative Medicine and Cell Therapy Industrialization Joint Project. This project unearths promising new “seeds,” mainly from start-up companies and researchers from within Kanagawa Prefecture, in the area of regenerative medicine and cell therapy. KSP, Inc. forms a joint project with the submitters of such proposals, and strives to promote the commercialization and industrialization of this field.
In addition to covering part of the expenses related to promoting the project (expenses leading to promotion of commercialization) it provides efficient and effective support by researching needs, opening sales channels etc.
Details of support
– Support for expenses in relation to the promotion of projects (expenses leading to promotion of commercialization)
– Hands-on support aimed at commercialization (researching needs, opening sales channels, preparing operating bases etc.)
Targeted projects
Projects implemented by companies (including those scheduled to be established) with a base for research and other operations within Japan, that fulfill all of the following criteria
– The project concerns regenerative medicine and cell therapy or related fields
– The project aims to carry out trial manufacture or commercialization of products or merchandise within Kanagawa Prefecture
– The project is worthy of special note for reasons of business feasibility or technological aspects
Flow of support
to pages of selected companies
Regenerative medicine & Cell therapy industrialization network of Kanagawa (RINK)
The Regenerative medicine & Cell therapy industrialization network of Kanagawa (RINK) was established to actively participate in and cooperate with a variety of related industry bodies, institutions, companies and academic institutions, primarily for the benefit of tenant companies of the Life Innovation Center, located on the King Skyfront.
The RINK’s goal is to promote cooperation and collaboration between companies and academic institutions etc. that are involved in the research and development or the commercialization of regenerative medicine and cell therapy, with the aim of accelerating the industrialization of the field.
Going forward, to achieve our ultimate goal to build a chain of processes (value chain) consisting of cell processing, cultivation, evaluation, storage and transport, we will work to expand the organization and to broaden and further energize its activities.










